Though there are a several ways that glass can be processed to make different products, there are two very distinct methods that have been practiced for many years – glass forming and glass molding. Of the two, glass forming has been used for hundreds of years. In very simple terms, it is taking a bubble of molten heated glass and blowing into it to form a drinking glass, vase, or pitcher. Read More…
S & S Optical has been leaders as glass fabricators in a variety of industries and markets for over 50 years. We offer a variety of different glasses to fit all of your needs. Also, we have a multitude of capabilities and services to offer-polishing, grinding, edging and more. To learn how we can fabricate your parts contact us today for a quote. We care about customers and quality service.
Glass cutting is a varied industry, and we have done it all for the past 50 years. We believe in manufacturing high quality glass products that will exceed customer expectations every time.
Founded in 1990, Technical Glass Products is a fabricator of Fused Quartz Glassware. We maintain a broad inventory of labware, flat stock, rod and tubes in addition to providing custom fabrication using state of the art equipment to suit your needs.
Coresix Precision Glass, Inc. is a fully integrated technical glass manufacturer specializing in high quality glass of all types. We are supported by a modern and fully integrated grinding department that can offer quality and cost effective manufacturing and fast delivery. Call us today for more information!
More Glass Forming Manufacturers
Glass comes in three different forms – soda lime, borosilicate, and phosphate. The two types that are used in glass forming are soda lime and borosilicate. Phosphate glass is a specialty product produced for industrial and scientific use. The type of glass we most commonly see is soda lime, which is the type that is made from silica sand. Borosilicate is used in glass forming to make cookware and heat resistant materials. Regardless of whether the glass is soda lime or borosilicate, the forming process is the same.
Glass forming begins with the combining of the raw materials. Soda lime glass is a combination of silica or sand, soda or sodium oxide, and lime or calcium oxide. When mixed, the largest amount, or 70%, of the mixture is silica. Soda is added to lower the melting temperature of the molten mix while lime stabilizes it. The mixing and heating is done in the batch housing where each step is carefully monitored and controlled.
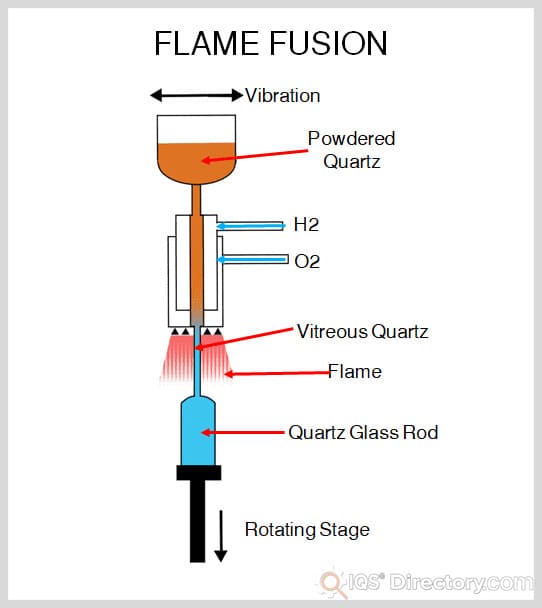
Once the mixture is completed and molten, it moves to the production center where it will be formed into different products. The production center consists of a set of molds. The batches from the batch housing are cut into little equal sized pieces called gobs. They are trimmed to the exact size needed for the product being produced. The gob has a puff of air forced into it to shape the parison, which is placed into the mold where it receives a stronger burst of air that forces it against the sides of the mold. Once formed, the molded product is allowed to cool before being moved to the finishing room.
The fully molded product is initially coated with a thin layer of metal and takes on the appearance of the finished product. When molded items cool, the outside that is exposed to the air cools faster than the inside creating an unevenness to the glass.
As with metal molds, glass is annealed, a process where the glass is heated and cooled multiple times until the inside and outside temperatures match.
The final stage of the process is the application of a polymer bath designed to give the exterior of the product a shiny finish and make it scratch resistant, increase its durability and strengthen it.
When glass has completed its use, it can be returned to a recycling center to be taken back to the production facility to be used in the creation of new glass products.





 Adhesives
Adhesives Alumina Ceramic
Alumina Ceramic Ceramic
Ceramic Glass
Glass Graphite
Graphite Lubricants
Lubricants Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services